
Cyclic Aza-peptide Integrin Ligand Synthesis and Biological Activity | The Journal of Organic Chemistry

Cyclic Aza-peptide Integrin Ligand Synthesis and Biological Activity | The Journal of Organic Chemistry

PDF) Productivity Improving of Artichoke in Organic Farming by Compost and Compost Tea Fertilization

Cyclic Aza-peptide Integrin Ligand Synthesis and Biological Activity | The Journal of Organic Chemistry

Dynamic Chirality in the Mechanism of Action of Allosteric CD36 Modulators of Macrophage-Driven Inflammation | Journal of Medicinal Chemistry
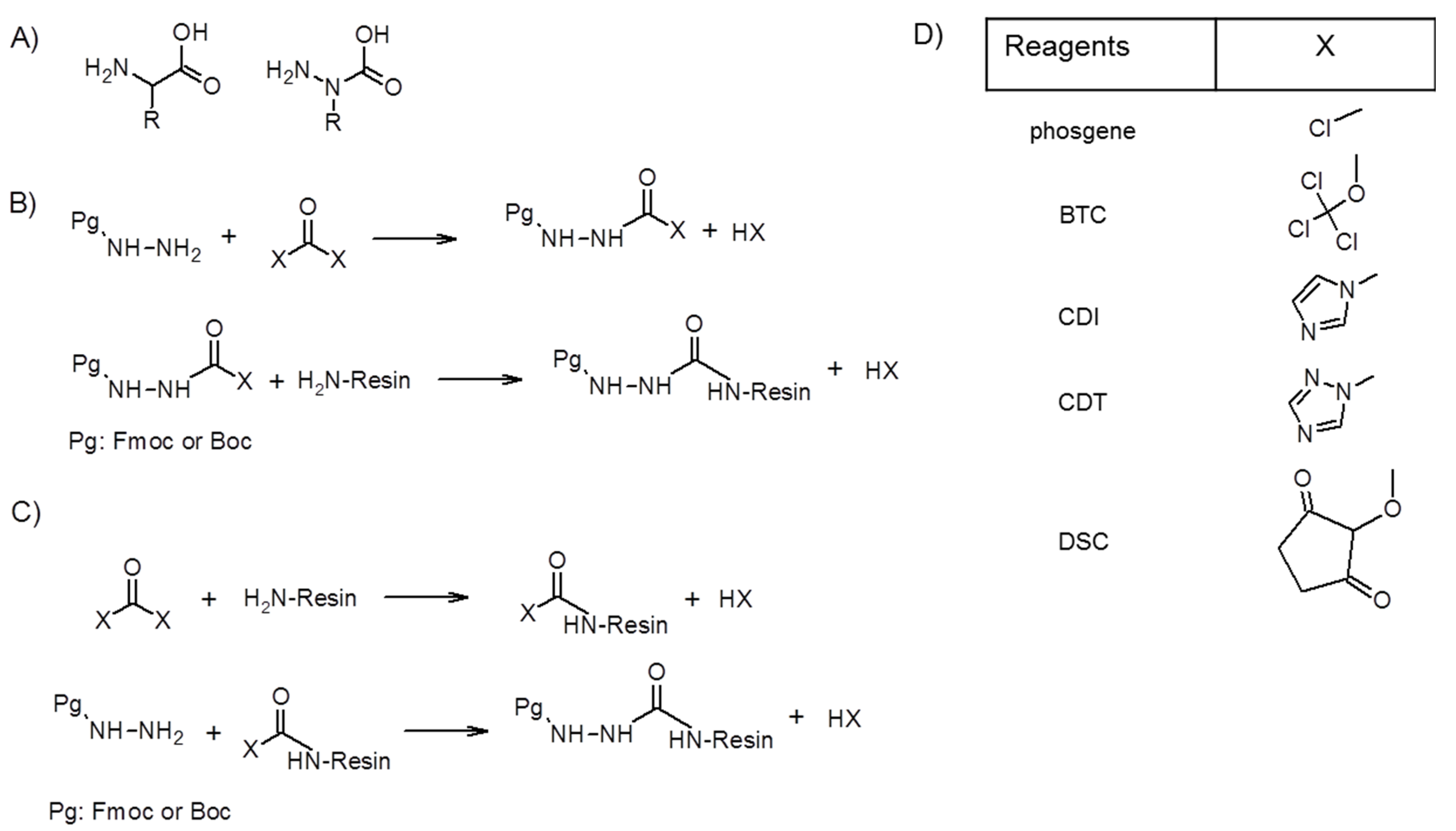
Future Pharmacology | Free Full-Text | Azapeptides as an Efficient Tool to Improve the Activity of Biologically Effective Peptides
top) Sequence representation of CMPs 6c, 14c, and 20c. (a) Graphical... | Download Scientific Diagram

Solid-Phase Azopeptide Diels–Alder Chemistry for Aza-pipecolyl Residue Synthesis To Study Peptide Conformation | The Journal of Organic Chemistry
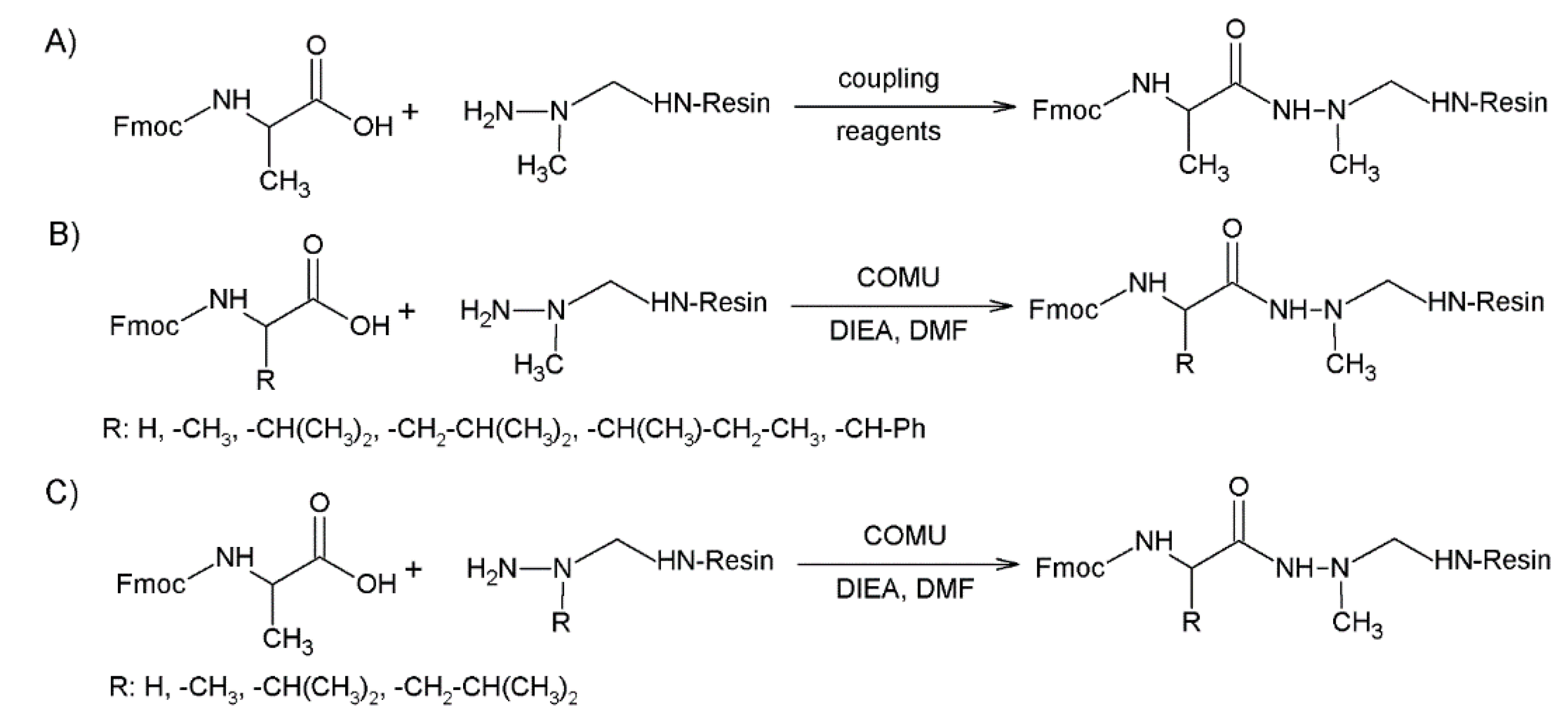
Future Pharmacology | Free Full-Text | Azapeptides as an Efficient Tool to Improve the Activity of Biologically Effective Peptides

Aza-Amino Acid Scan for Rapid Identification of Secondary Structure Based on the Application of N-Boc-Aza1-Dipeptides in Peptide Synthesis | Journal of the American Chemical Society

Solid-Phase Azopeptide Diels–Alder Chemistry for Aza-pipecolyl Residue Synthesis To Study Peptide Conformation | The Journal of Organic Chemistry

Future Pharmacology | Free Full-Text | Azapeptides as an Efficient Tool to Improve the Activity of Biologically Effective Peptides

Aza-Amino Acid Scanning of Secondary Structure Suited for Solid-Phase Peptide Synthesis with Fmoc Chemistry and Aza-Amino Acids with Heteroatomic Side Chains | ACS Combinatorial Science









