
Recent Advances of the Halogen–Zinc Exchange Reaction - Balkenhohl - 2020 - Chemistry – A European Journal - Wiley Online Library

Consecutive Double 1,2‐Migration of Two Different Groups in Silyl(dichloromethyl)cuprates - Kondo - 2004 - Angewandte Chemie - Wiley Online Library

An Evaluation of Multiple Catalytic Systems for the Cyanation of 2,3-Dichlorobenzoyl Chloride: Application to the Synthesis of Lamotrigine | Organic Process Research & Development

Regioselective and Stereoselective Copper(I)-Promoted Allylation and Conjugate Addition of N-Boc-2-lithiopyrrolidine and N-Boc-2-lithiopiperidine | The Journal of Organic Chemistry

Cobalt(II)-Catalyzed Cross-Coupling between Polyfunctional Arylcopper Reagents and Aryl Fluorides or Tosylates | Organic Letters
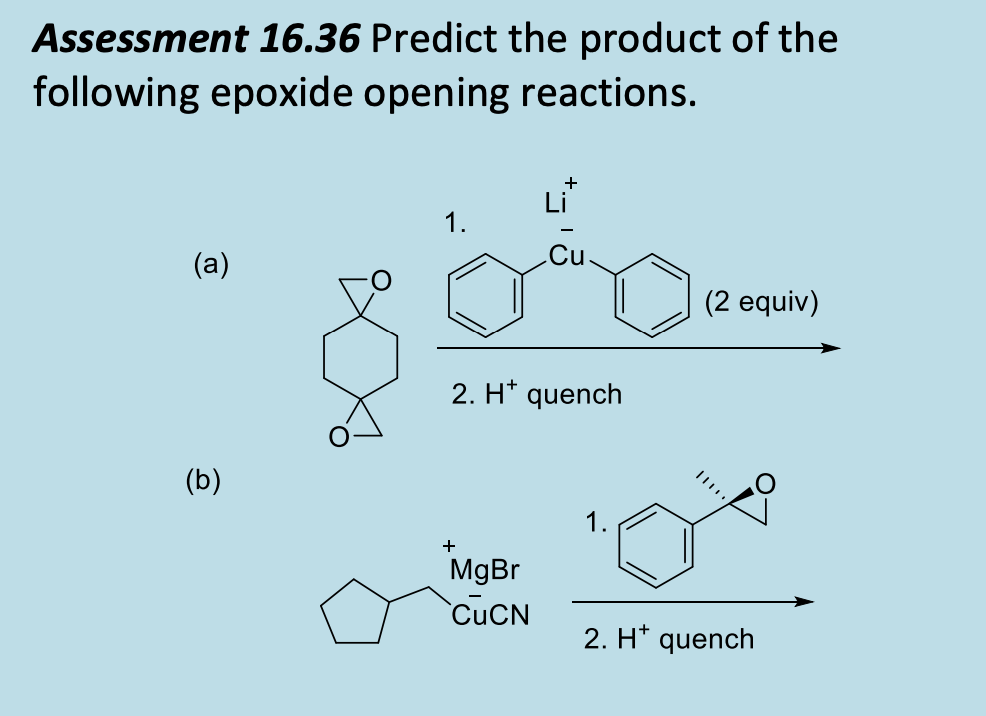
![What is the product (assume acidic quench)? What is the product? [{Image src='rection1918034682601693189.jpg' alt='reaction' caption=''}] | Homework.Study.com What is the product (assume acidic quench)? What is the product? [{Image src='rection1918034682601693189.jpg' alt='reaction' caption=''}] | Homework.Study.com](https://homework.study.com/cimages/multimages/16/rection1918034682601693189.jpg)
What is the product (assume acidic quench)? What is the product? [{Image src='rection1918034682601693189.jpg' alt='reaction' caption=''}] | Homework.Study.com

Copper-Catalyzed Recycling of Halogen Activating Groups via 1,3-Halogen Migration | Journal of the American Chemical Society
![Structure of [(CuCN) 3 (bdpzmpy)] and its guest-dependent luminescence... | Download Scientific Diagram Structure of [(CuCN) 3 (bdpzmpy)] and its guest-dependent luminescence... | Download Scientific Diagram](https://www.researchgate.net/publication/298899651/figure/fig2/AS:347011535654916@1459745405883/Structure-of-CuCN-3-bdpzmpy-and-its-guest-dependent-luminescence-induced-by-change.png)
Structure of [(CuCN) 3 (bdpzmpy)] and its guest-dependent luminescence... | Download Scientific Diagram

New In Situ Trapping Metalations of Functionalized Arenes and Heteroarenes with TMPLi in the Presence of ZnCl2 and Other Metal Salts - Frischmuth - 2014 - Angewandte Chemie International Edition - Wiley Online Library
![PDF] Polyfunctionalizations of N-Heterocycles via Chemo- and Regioselective Metalations | Semantic Scholar PDF] Polyfunctionalizations of N-Heterocycles via Chemo- and Regioselective Metalations | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/6d41aec601614682d38918893fe1baf69032dce1/43-Table1-1.png)
PDF] Polyfunctionalizations of N-Heterocycles via Chemo- and Regioselective Metalations | Semantic Scholar

Scheme 12 Transmetallation of organozinc reagent 31 with CuCN·2LiCl to... | Download Scientific Diagram
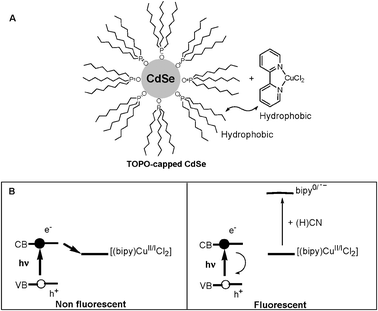
Selective turn-on fluorescence detection of cyanide in water using hydrophobic CdSe quantum dots - Chemical Communications (RSC Publishing) DOI:10.1039/B716194H

Scheme 8 Generation and possible products from aqueous quenching of... | Download Scientific Diagram

Copper Cyanide-Catalyzed Palladium Coupling of N-tert-Butoxycarbonyl-Protected α-Lithio Amines with Aryl Iodides or Vinyl Iodides | The Journal of Organic Chemistry

Scheme 12 Transmetallation of organozinc reagent 31 with CuCN·2LiCl to... | Download Scientific Diagram

The Renaissance of Organo Nitriles in Organic Synthesis - Rakshit - 2022 - Chemistry – An Asian Journal - Wiley Online Library

Copper(I) Cyanide Networks: Synthesis, Luminescence Behavior and Thermal Analysis. Part 1. Diimine Ligands | Inorganic Chemistry

Regioselective and Stereoselective Copper(I)-Promoted Allylation and Conjugate Addition of N-Boc-2-lithiopyrrolidine and N-Boc-2-lithiopiperidine | The Journal of Organic Chemistry

Scheme 57 Reagents: i, (COCl) 2 ; ii, CuCN; iii, H 2 , Pd/C, AcOH; iv,... | Download Scientific Diagram
Scheme 7. Reagents and conditions: (a) NaNO 2 , pTSA, KI, tBuOH, H 2 O,... | Download Scientific Diagram

Regioselective and Stereoselective Copper(I)-Promoted Allylation and Conjugate Addition of N-Boc-2-lithiopyrrolidine and N-Boc-2-lithiopiperidine | The Journal of Organic Chemistry

Copper(I) Cyanide Networks: Synthesis, Luminescence Behavior and Thermal Analysis. Part 1. Diimine Ligands | Inorganic Chemistry

![3D network of [(CuCN)3(L1)]n Perspective view down the a axis | Download Scientific Diagram 3D network of [(CuCN)3(L1)]n Perspective view down the a axis | Download Scientific Diagram](https://www.researchgate.net/publication/303090649/figure/fig21/AS:961779484090368@1606317506005/3D-network-of-CuCN3L1n-Perspective-view-down-the-a-axis.gif)