Question Video: Calculating a Cell Potential from Standard Electrode Potentials of Cadmium and Nickel | Nagwa

Standard reduction potential of different species in aqueous solution... | Download Scientific Diagram
![Expert Answer] calculate standard electrode potential of Ni/Ni2+,if cell potential of the - Brainly.in Expert Answer] calculate standard electrode potential of Ni/Ni2+,if cell potential of the - Brainly.in](https://hi-static.z-dn.net/files/d51/b7359f4685f931af8cd83cf0636289d3.jpg)
Expert Answer] calculate standard electrode potential of Ni/Ni2+,if cell potential of the - Brainly.in

Calculate the standard electrode potential of Ni2+/Ni electrode if |Class 12 CHEMISTRY | Doubtnut - YouTube

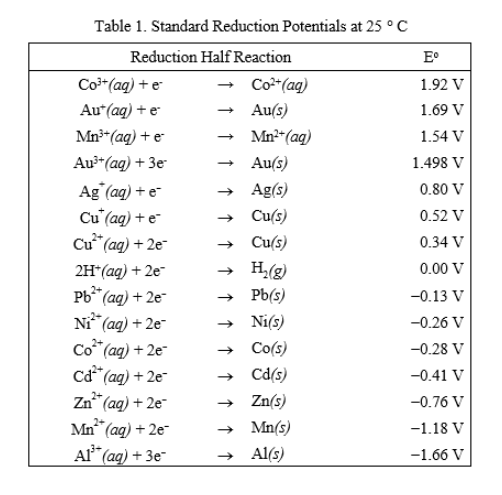
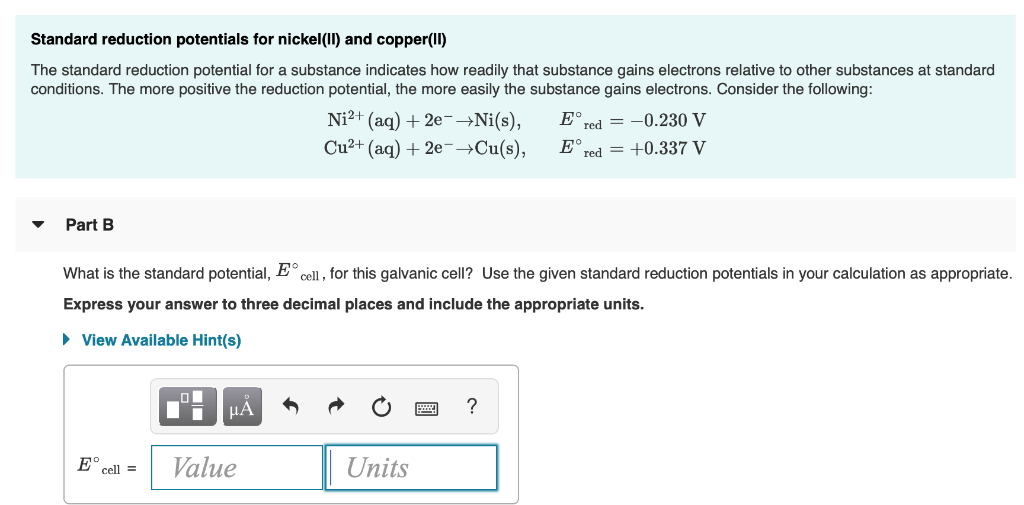
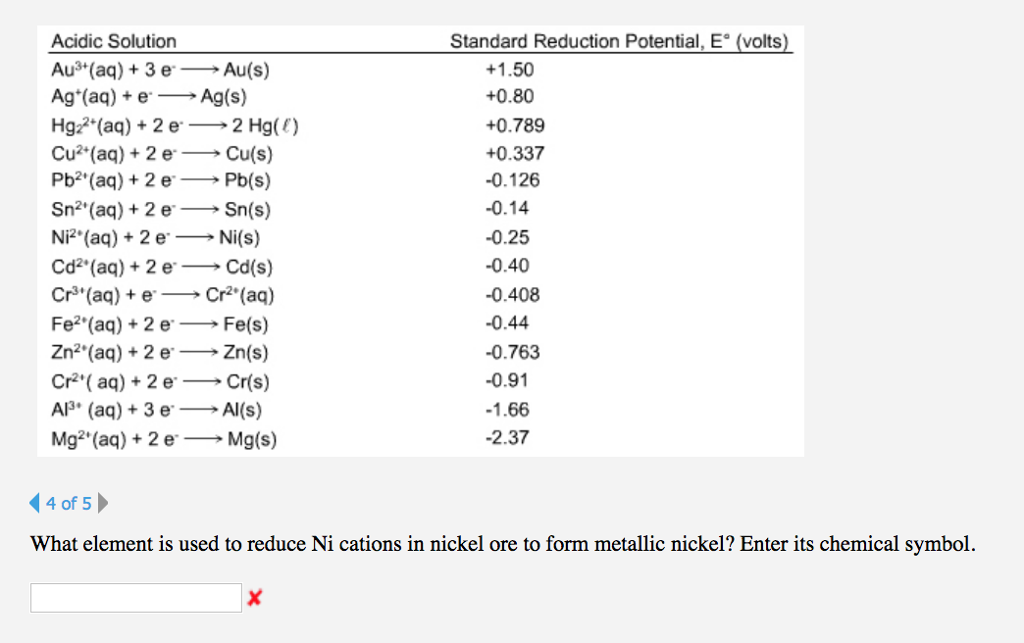
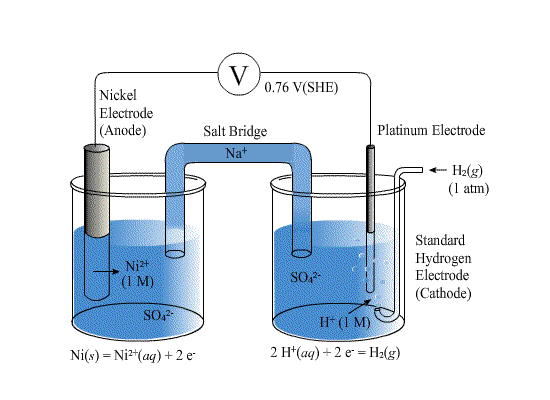
The standard electrode potentials of Zn and Ni respectively are - 0.76 V and - 0.25 V. Then the standard emf of the spontaneous cell by coupling these under standard conditions is:

Table 4 from Nickel(II) complexes with tetra- and pentadentate aminopyridine ligands: synthesis, structure, electrochemistry, and reduction to nickel(I) species. | Semantic Scholar

Table 1 from Co-electrodeposited Mesoporous PtM ( M = Co , Ni , Cu ) as an Active Catalyst for Oxygen Reduction Reaction in a PEMFC | Semantic Scholar

Standard Reduction Potential (E): when given two half reactions and then asked to give E for a reac… | Reduction potential, Chemistry education, Across the universe

Calculate the standard electrode potential of the Ni^(2+)//Ni electrode , if the cell potential potential of the cell, Ni//N^(2+)(0.01 M)//Cu is 0.59" V ". "Given" E(Cu^(2+)//Cu)^(@)=+0.34 " V "