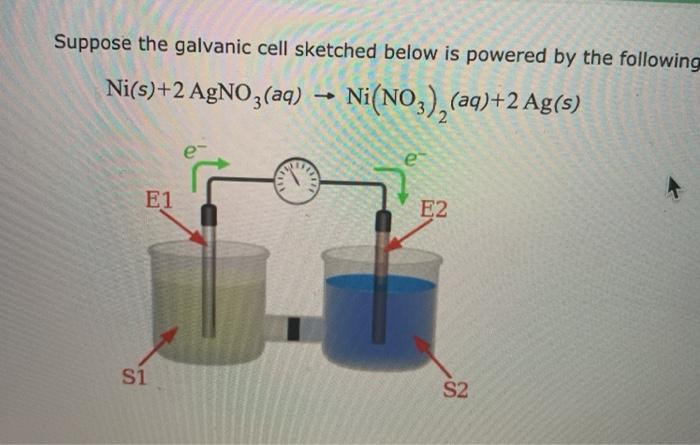
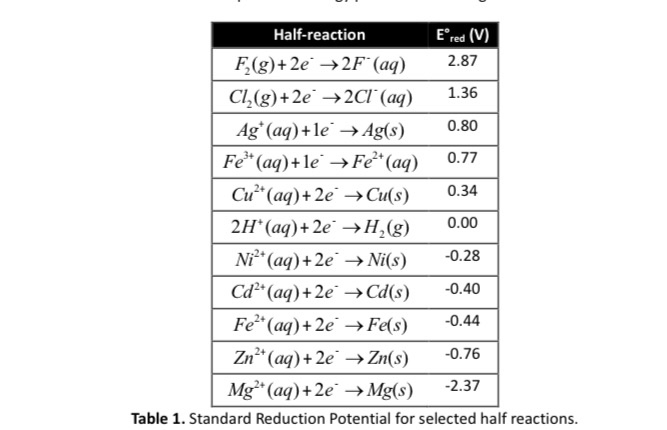
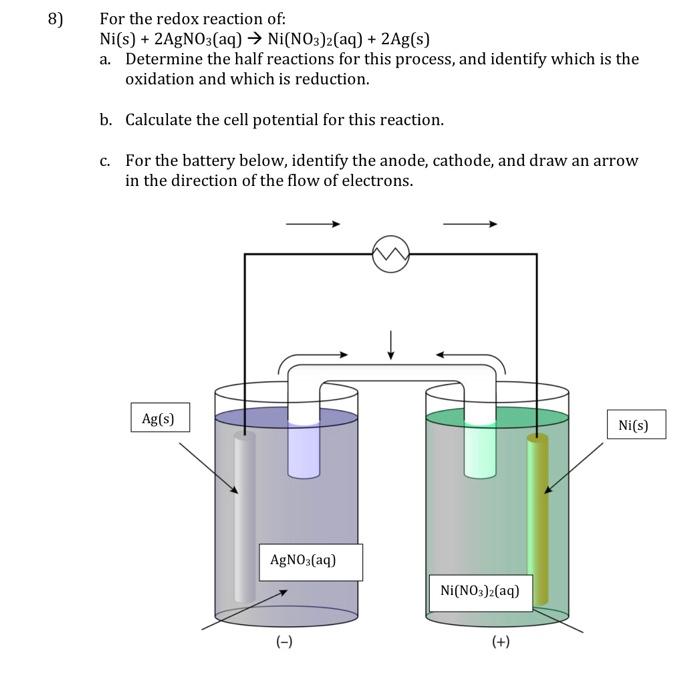
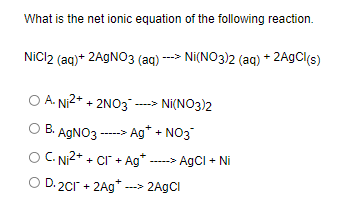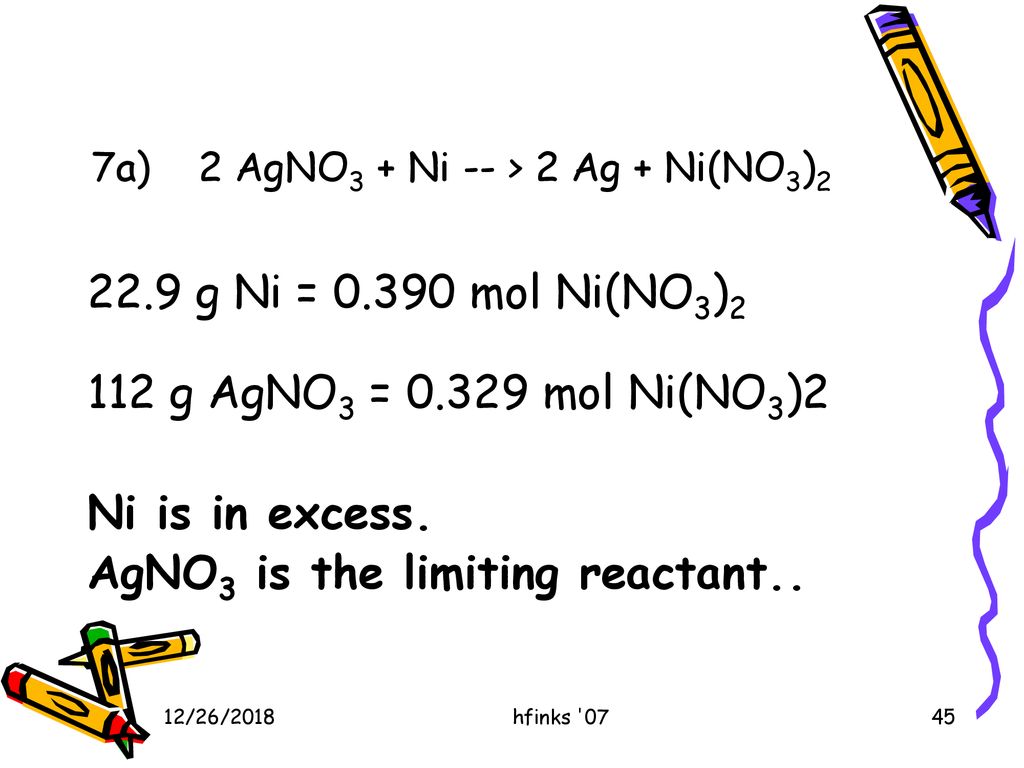A strip of nickel metal is placed in a 1 molar solution of Ni(NO3)2 and a strip of silver metal - Chemistry - Electrochemistry - 9165083 | Meritnation.com

OneClass: check the results of your tests against the solubility rules and write balanced equation fo...

SOLVED: Select the correct balanced net ionic equation for each of the following reactions occuring in aquequssolution Cu(NO3h2 Ni(NO3)2 AgNO3 NaCl Agcl NaNO3 Ca(NO3h2 KzCo3 Caco3 2KNO3 Cuz+ 2NO3 Ni(NO3)2 Ag+ NaNO3