
Cr2O7 + SO32- gives Cr3+ + SO42-. how to balance this equation by oxidation number method? | EduRev Class 11 Question

Balance the following redox reaction. (Cr2O7)(aq)^2 - + Fe(aq)^2 + → Cr(aq)^3 + + Fe(aq)^3 + (acidic medium)

For the redox reaction Cr(2)O(7)^(-2)+H^(+)+Ni rarr Cr^(3)+Ni^(2+)+H(2)O The correct coefficients of the reactions for the balanced reaction are

The configurations of H2CrO4 and Cr2O7 2-adsorption on the N, O, P or... | Download Scientific Diagram
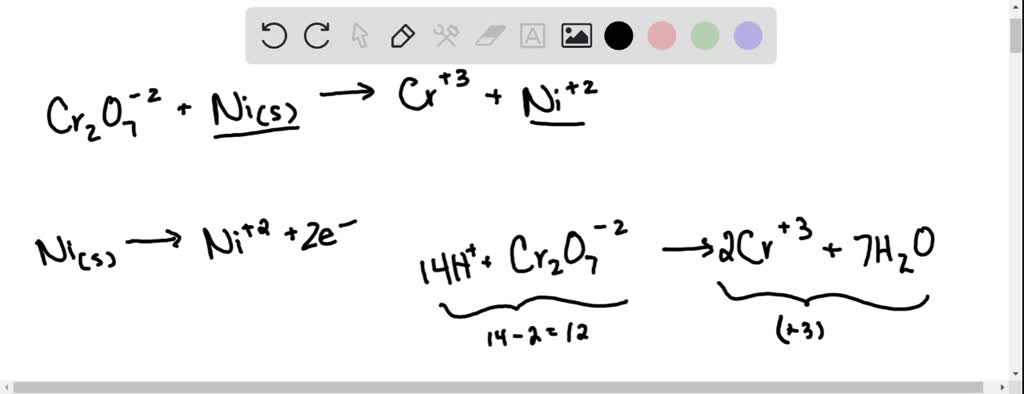
SOLVED: Balance the following redox equation in an acidic solution : Cr2O7 -2 (aq) + Ni (s) —– > Ni2+ (aq) + Cr3+ (aq) (acidic solution)
![Novel mode of 2-fold interpenetration observed in a primitive cubic network of formula [Ni(1,2-bis(4-pyridyl)acetylene)2(Cr2O7)]n - Chemical Communications (RSC Publishing) Novel mode of 2-fold interpenetration observed in a primitive cubic network of formula [Ni(1,2-bis(4-pyridyl)acetylene)2(Cr2O7)]n - Chemical Communications (RSC Publishing)](https://pubs.rsc.org/en/Content/Image/GA/C5CC05866J)
Novel mode of 2-fold interpenetration observed in a primitive cubic network of formula [Ni(1,2-bis(4-pyridyl)acetylene)2(Cr2O7)]n - Chemical Communications (RSC Publishing)
Balance the following ionic equations (i) Cr2O7^2-+H^++I^- → Cr^3+ +I2+H2O - Sarthaks eConnect | Largest Online Education Community

SOLVED: 16.Consider the reaction Cr2O7^2- + 6 S2O3^2- + 14 H^+ ↔ 2 Cr^3+ + 3 S4O6^2- + 7 H2O. What is the oxidizing agent? * S2O3^2- S4O6^2- Cr^3+ Cr2O7^2- 17.It is
Balance the following ionic equations (i) Cr2O7^2-+H^++I^- → Cr^3+ +I2+H2O - Sarthaks eConnect | Largest Online Education Community

For the redox reaction Cr(2)O(7)^(-2)+H^(+)+Ni rarr Cr^(3)+Ni^(2+)+H(2)O The correct coefficients of the reactions for the balanced reaction are





![Polar Compounds Constructed with the [Cr2O7]2- Anion | Inorganic Chemistry Polar Compounds Constructed with the [Cr2O7]2- Anion | Inorganic Chemistry](https://pubs.acs.org/cms/10.1021/ic000953y/asset/images/medium/ic000953yn00001.gif)




![Polar Compounds Constructed with the [Cr2O7]2- Anion | Inorganic Chemistry Polar Compounds Constructed with the [Cr2O7]2- Anion | Inorganic Chemistry](https://pubs.acs.org/cms/10.1021/ic000953y/asset/images/large/ic000953yn00001.jpeg)


![Ni(H2O)6]2(Cr2O7)2(hmta)4•2H2О (HMTA - hexamethylenetetramine) : r/chemistry Ni(H2O)6]2(Cr2O7)2(hmta)4•2H2О (HMTA - hexamethylenetetramine) : r/chemistry](https://i.redd.it/c6l3dusld2471.jpg)