Synthesis Of Tris Ethylenediamine Nickel Ii Chloride Lab Report - Fill Online, Printable, Fillable, Blank | pdfFiller

Nickel Complexes of a Pincer NN2 Ligand: Multiple Carbon−Chloride Activation of CH2Cl2 and CHCl3 Leads to Selective Carbon−Carbon Bond Formation | Journal of the American Chemical Society

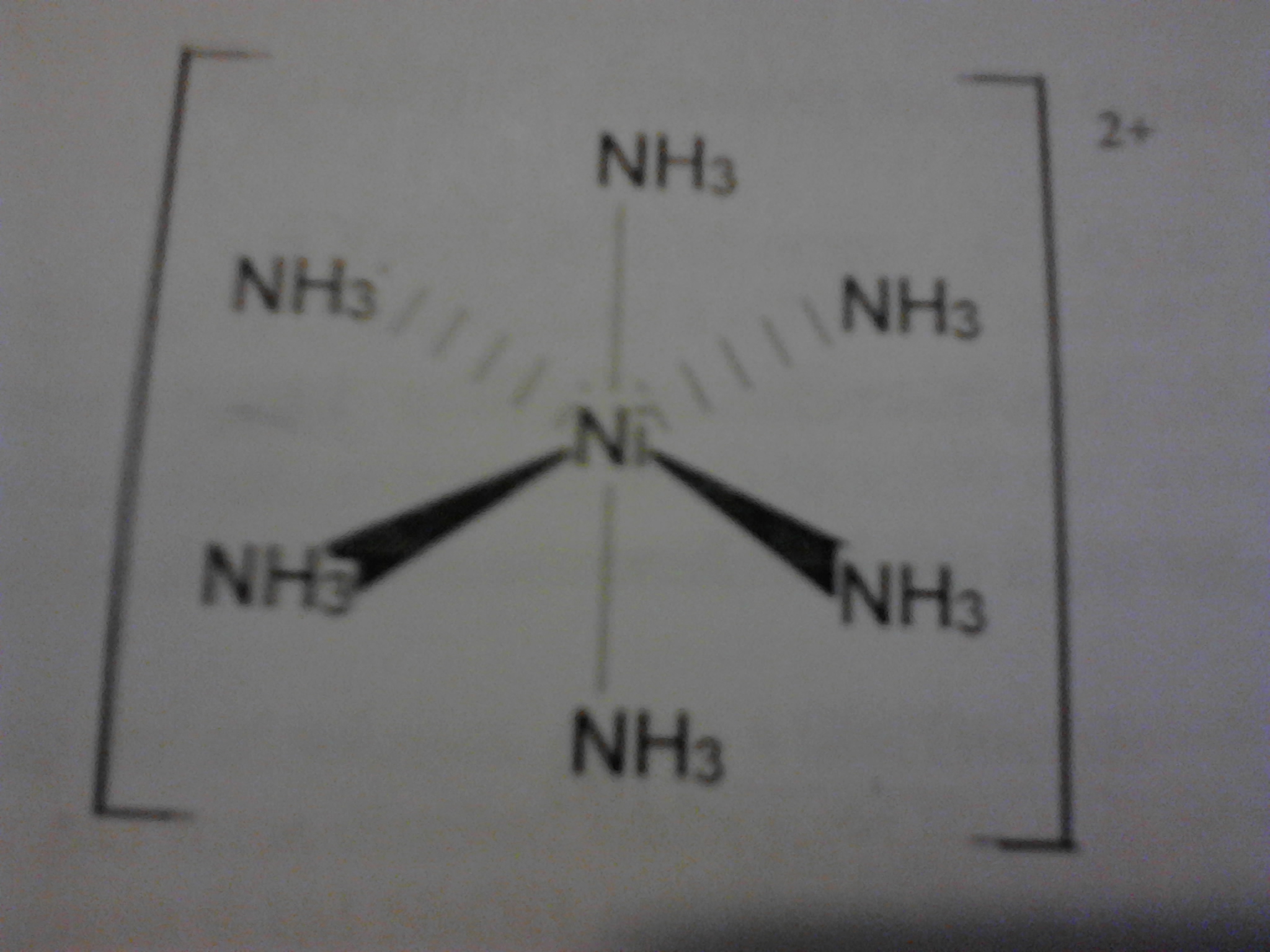
This is hexaammine nickel II chloride. This is a complex of ammonia formed with nickel II Chloride which has a beautiful purple color .It's really unstable and on sitting for few days

Nickel transition metal Chemistry nickel(II) Ni2+ complex ions ligand substitution redox chemical reactions principal oxidation states +2 +3 GCE AS A2 IB A level inorganic chemistry revision notes

SOLVED: 3 . Synthesis of tris(ethylenediamine)nickel(II) chloride hydrate Tris(ethylenediamine)nickel(Il) chloride dilydrate Three ethylenediamine (en) molecules displace the six water ligands of [Ni(HzOJJCIz to fomm violet tris(ethylenediamine)nickel ...
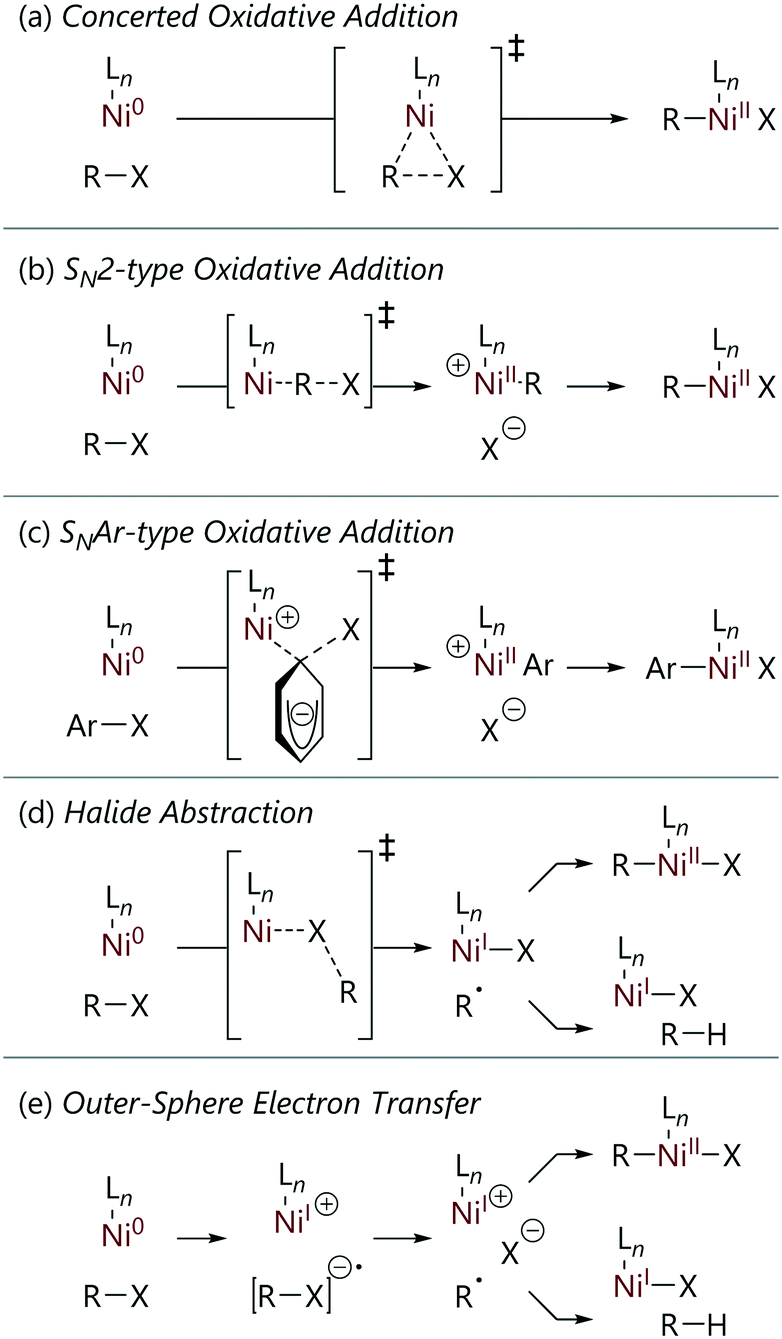
Reactions of nickel(0) with organochlorides, organobromides, and organoiodides: mechanisms and structure/reactivity relationships - Catalysis Science & Technology (RSC Publishing) DOI:10.1039/D1CY00374G

Stable Nickel(I) Complexes with Electron-Rich, Sterically-Hindered, Innocent PNP Pincer Ligands | Organometallics

Nickel transition metal Chemistry nickel(II) Ni2+ complex ions ligand substitution redox chemical reactions principal oxidation states +2 +3 GCE AS A2 IB A level inorganic chemistry revision notes