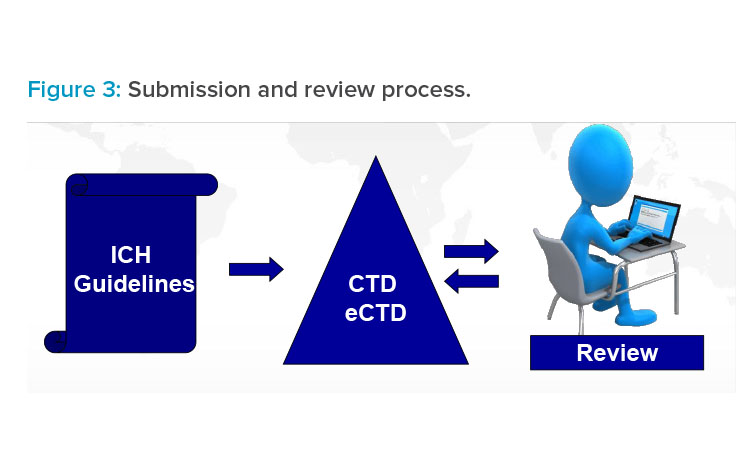
International Conference on Harmonisation (ICH) Quality Guidelines: Pharmaceutical, Biologics, and Medical Device Guidance Documents Concise Reference: 9780982147658: Medicine & Health Science Books @ Amazon.com

The Good Clinical Practice guideline and its interpretation – perceptions of clinical trial teams in sub‐Saharan Africa - Vischer - 2016 - Tropical Medicine & International Health - Wiley Online Library
![ROLE OF ICH GUIDELINES FROM ICH-Q1 to ICH-Q14 by Rajashri Ojha[Founder & Director Raaj GPRAC] - YouTube ROLE OF ICH GUIDELINES FROM ICH-Q1 to ICH-Q14 by Rajashri Ojha[Founder & Director Raaj GPRAC] - YouTube](https://i.ytimg.com/vi/KsS_vr312Lo/maxresdefault.jpg?sqp=-oaymwEmCIAKENAF8quKqQMa8AEB-AHUBoAC4AOKAgwIABABGGUgZShlMA8=&rs=AOn4CLAvPIP2OugROMUZz62Y_9En20YfSw)
ROLE OF ICH GUIDELINES FROM ICH-Q1 to ICH-Q14 by Rajashri Ojha[Founder & Director Raaj GPRAC] - YouTube

Origin and principles of international conference on harmonization- Good clinical practices (ICH-GCP)) | PPT

Stream )% International Conference on Harmonisation, ICH Quality Guidelines, Pharmaceutical, Biologics by User 669825415 | Listen online for free on SoundCloud



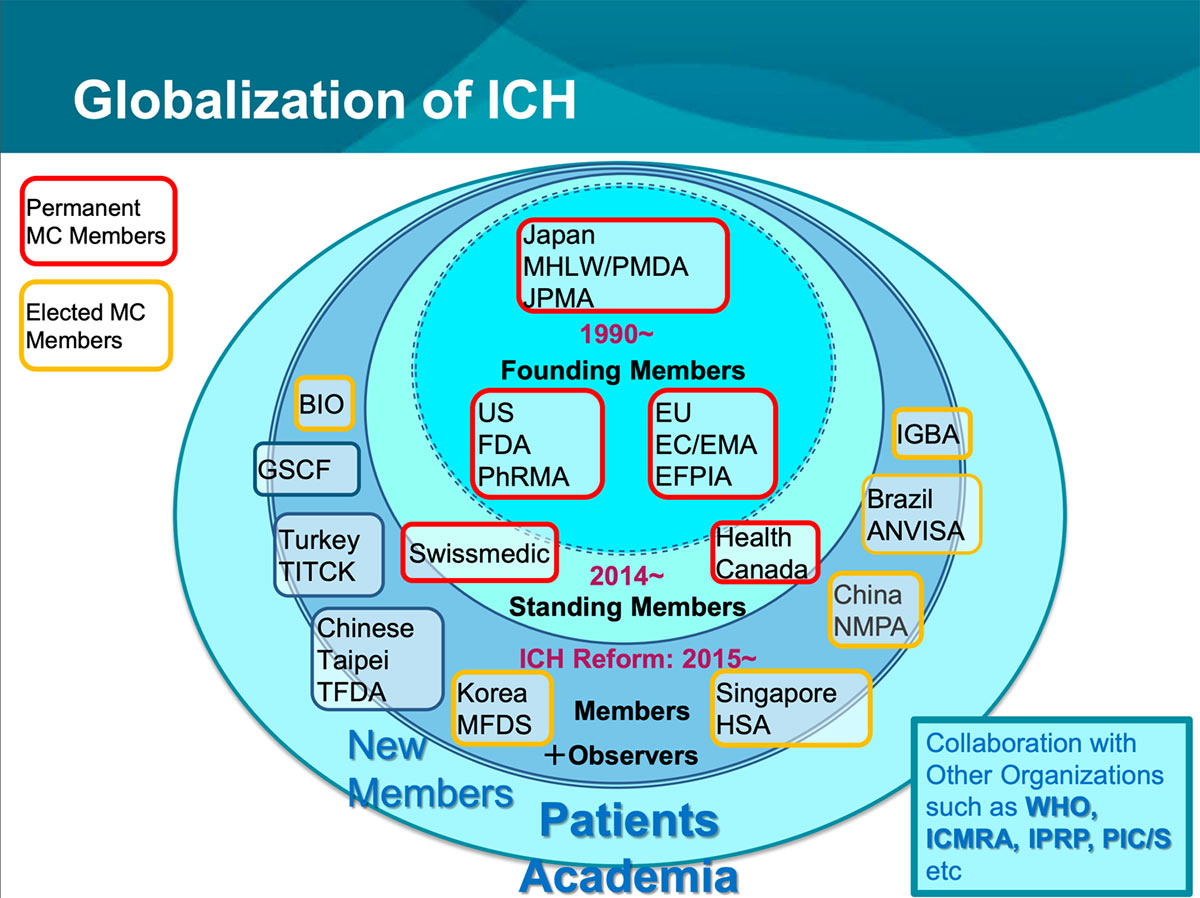

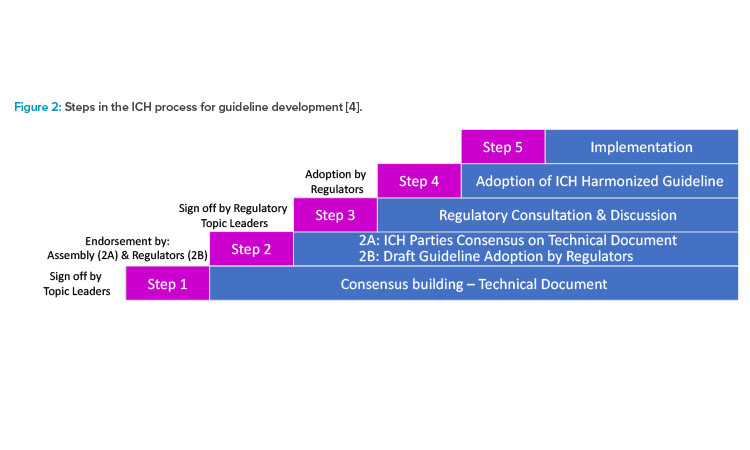









(135).jpg)