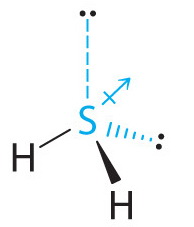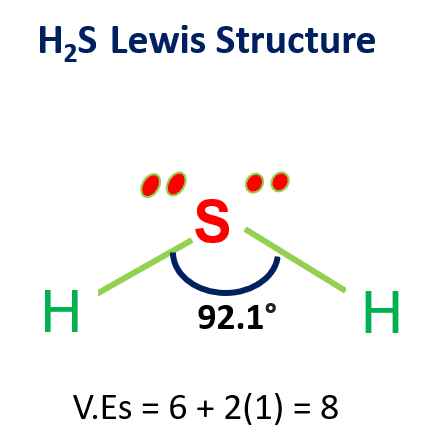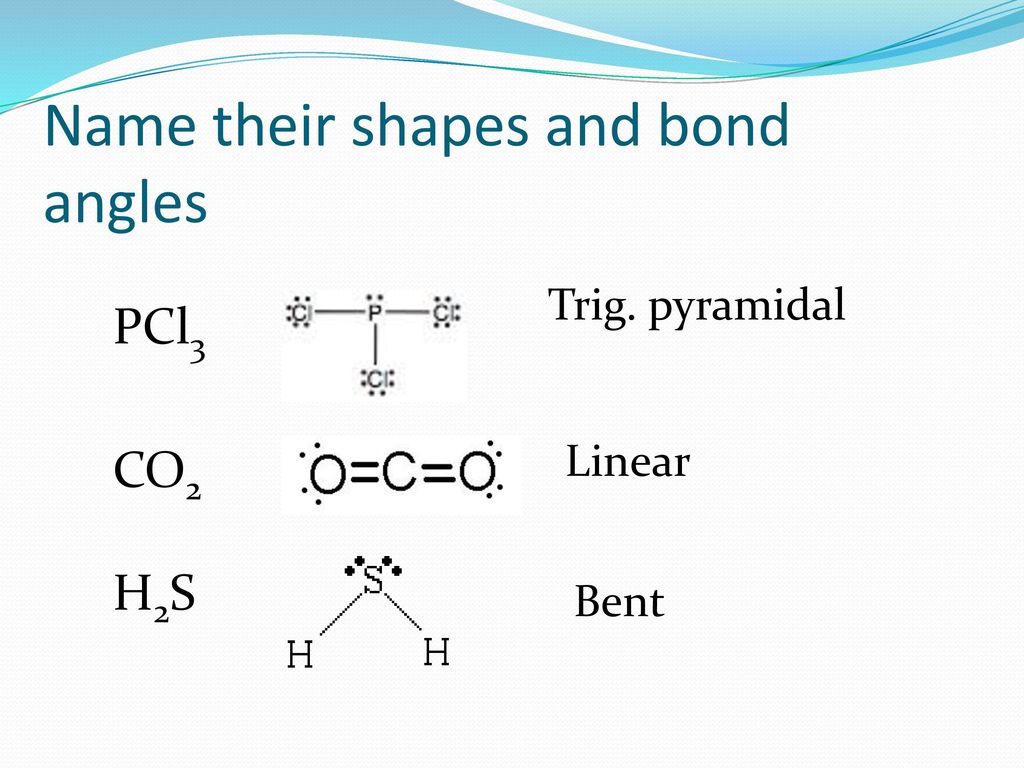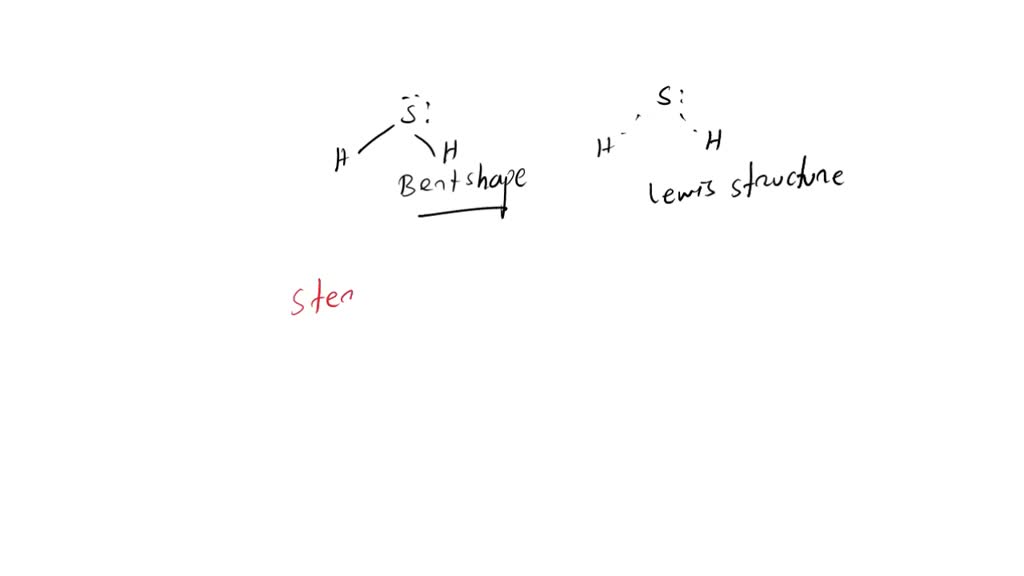
SOLVED: Which option best describes hydrogen sulfide H2S, according to its electronic, molecular shape and polarity, respectively. Draw the Lewis structure for the compound. Group of answer choices Trigonal planar, trigonal planar,

H2S Molecular Geometry | Shape and Bond Angles (Hydrogen Sulphide) | H2S Molecular Geometry | Shape and Bond Angles (Hydrogen Sulphide) H2S is a chemical formula of Hydrogen Sulphide gas. It is

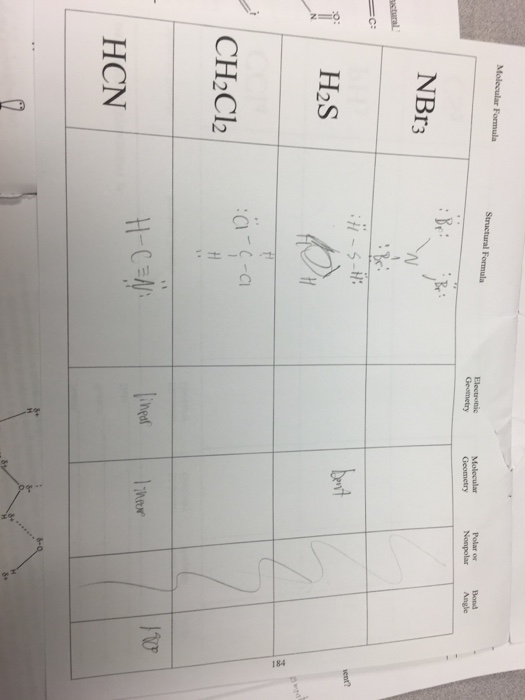
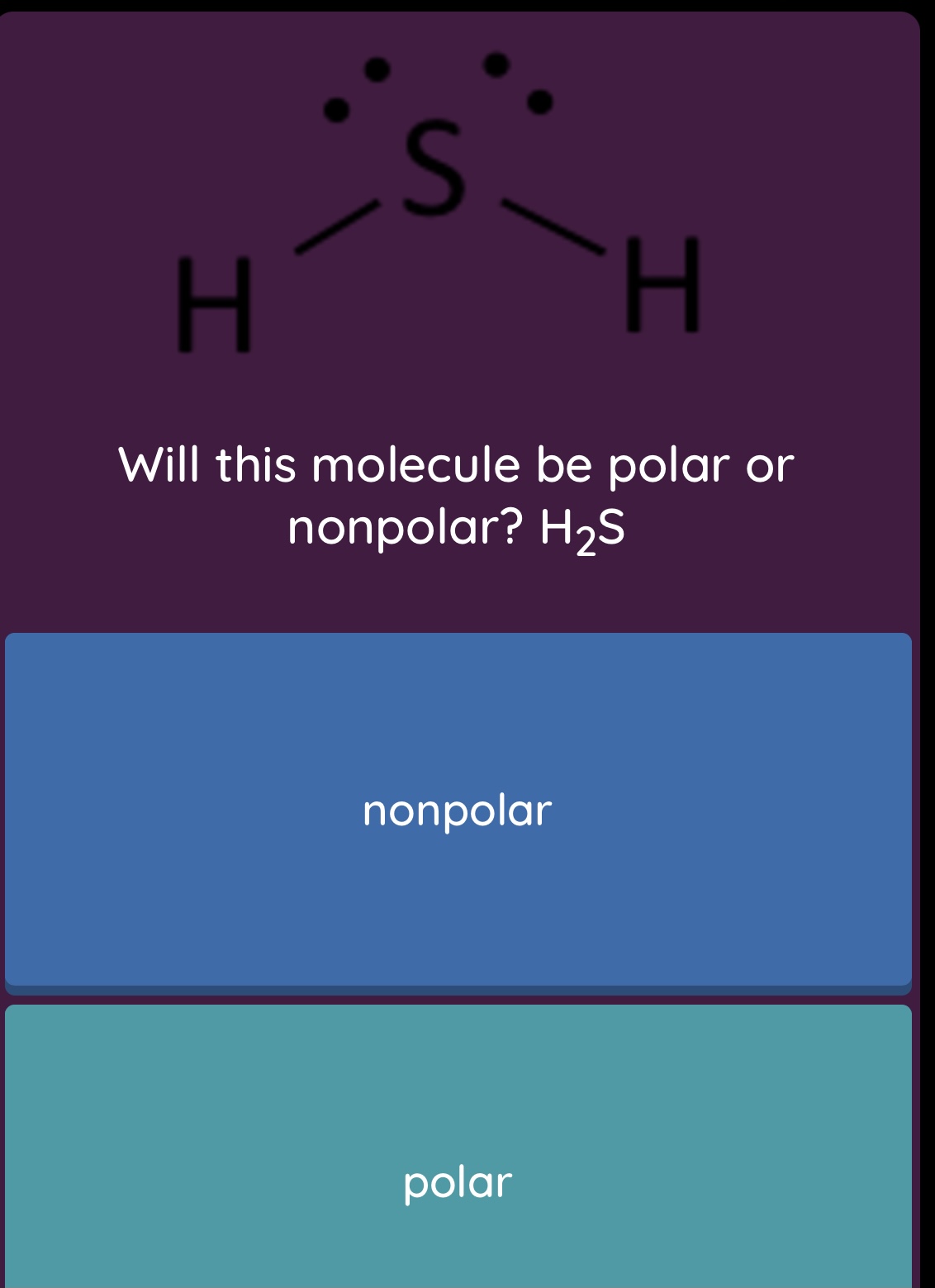
SOLVED: molecule or polyatomic ion polar or atom closest to nonpolar? negative side polar nonpolar polar nonpolar polar nonpolar H2S CH;F Cl2



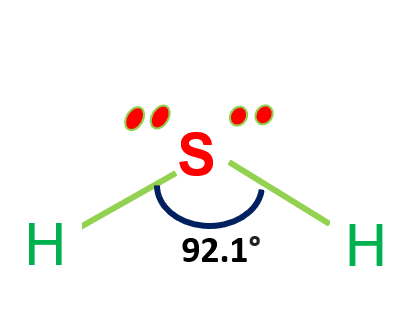
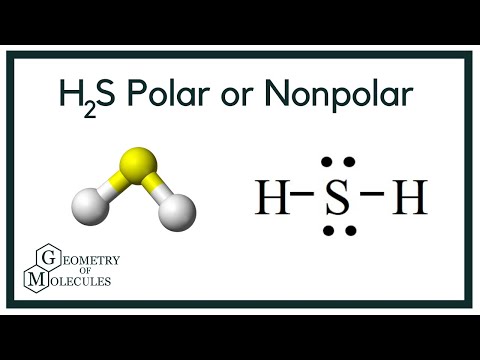
![Is [math]H_2S[/math] polar or nonpolar? - Quora Is [math]H_2S[/math] polar or nonpolar? - Quora](https://qph.cf2.quoracdn.net/main-qimg-60b021aa16478c549d60c016b6a83f1f.webp)