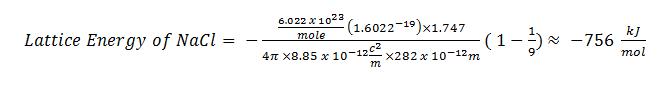Tutorial Week 6 Answers 2018 - Warning: TT: undefined function: 32 Tutorial Week 6 Reactions and - Studocu
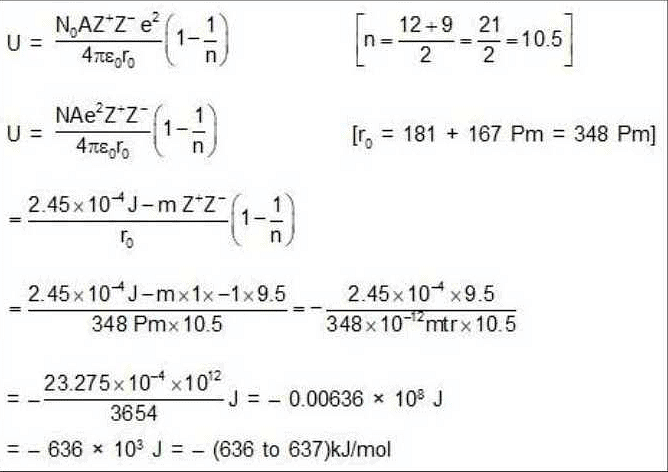
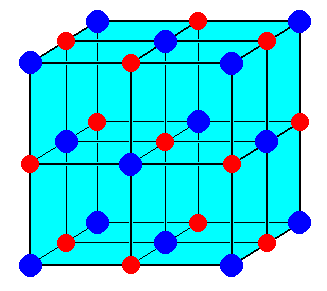
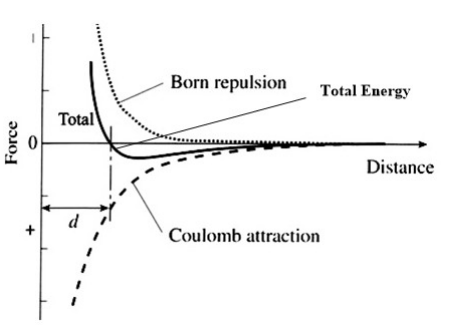
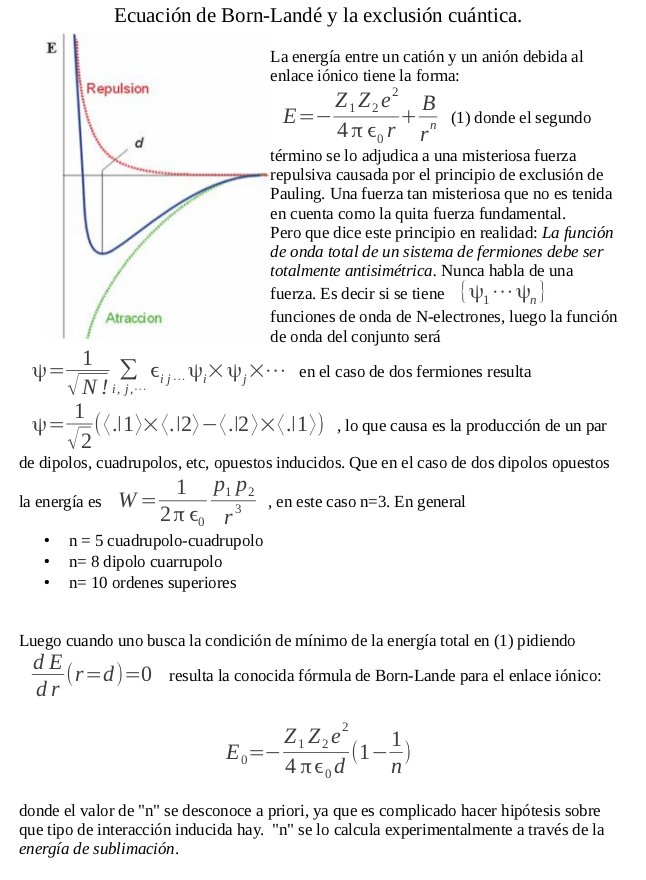
The ionic radii of Cs+ and Cl- ions are 181 and 167 pm, respectively. The Born exponents for the He, Ne, Ar, Kr and Xe configurations are 5, 7, 9, 10 and