BC Cancer Protocol Summary for Treatment of Relapsed or Refractory Pre-B Cell Acute Lymphoblastic Leukemia with Inotuzumab Ozoga

PDF) Cap 1 Messenger RNA Synthesis with Co-transcriptional CleanCap® Analog by In Vitro Transcription

Camrelizumab plus apatinib as second-line treatment for advanced oesophageal squamous cell carcinoma (CAP 02): a single-arm, open-label, phase 2 trial - The Lancet Gastroenterology & Hepatology

Rationale and Design of the Targeted Agent and Profiling Utilization Registry Study | JCO Precision Oncology
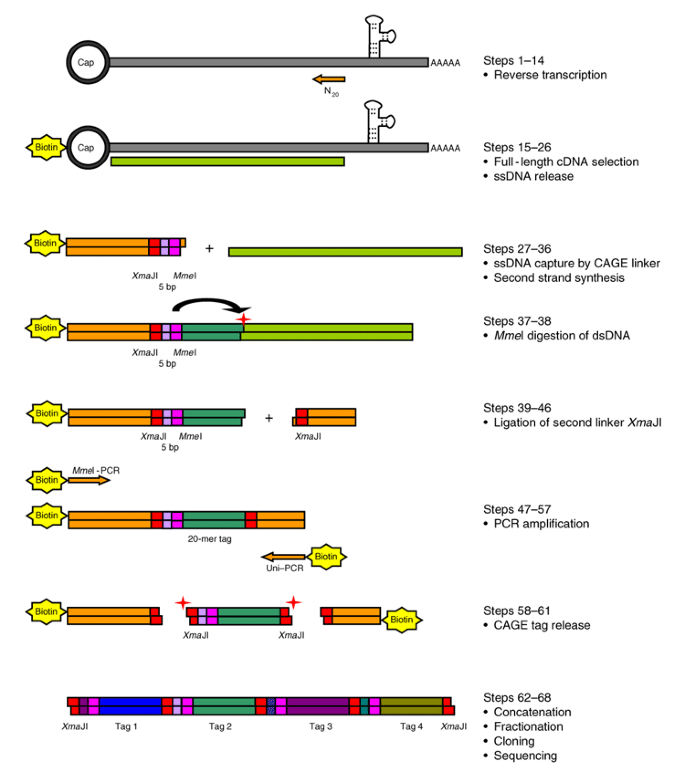
Automated Workflow for Preparation of cDNA for Cap Analysis of Gene Expression on a Single Molecule Sequencer | PLOS ONE
J2G-GH-JZJK Protocol(b) A Phase 2 Study of Oral Selpercatinib (LOXO-292) in Patients with Advanced Solid Tumors, Including RET F

Direct identification of ALK and ROS1 fusions in non-small cell lung cancer from hematoxylin and eosin-stained slides using deep learning algorithms | Modern Pathology

Cap 1 Messenger RNA Synthesis with Co‐transcriptional CleanCap® Analog by In Vitro Transcription - Henderson - 2021 - Current Protocols - Wiley Online Library

CAP-MAP: cap analysis protocol with minimal analyte processing, a rapid and sensitive approach to analysing mRNA cap structures | Open Biology
Protocol for the Examination of Resection Specimens From Patients With Primary Non-Small Cell Carcinoma, Small Cell Carcinoma, o