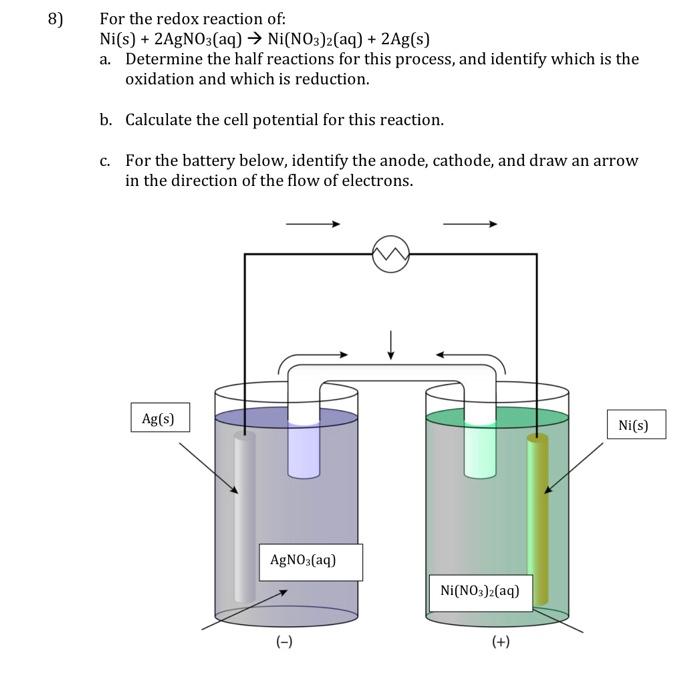
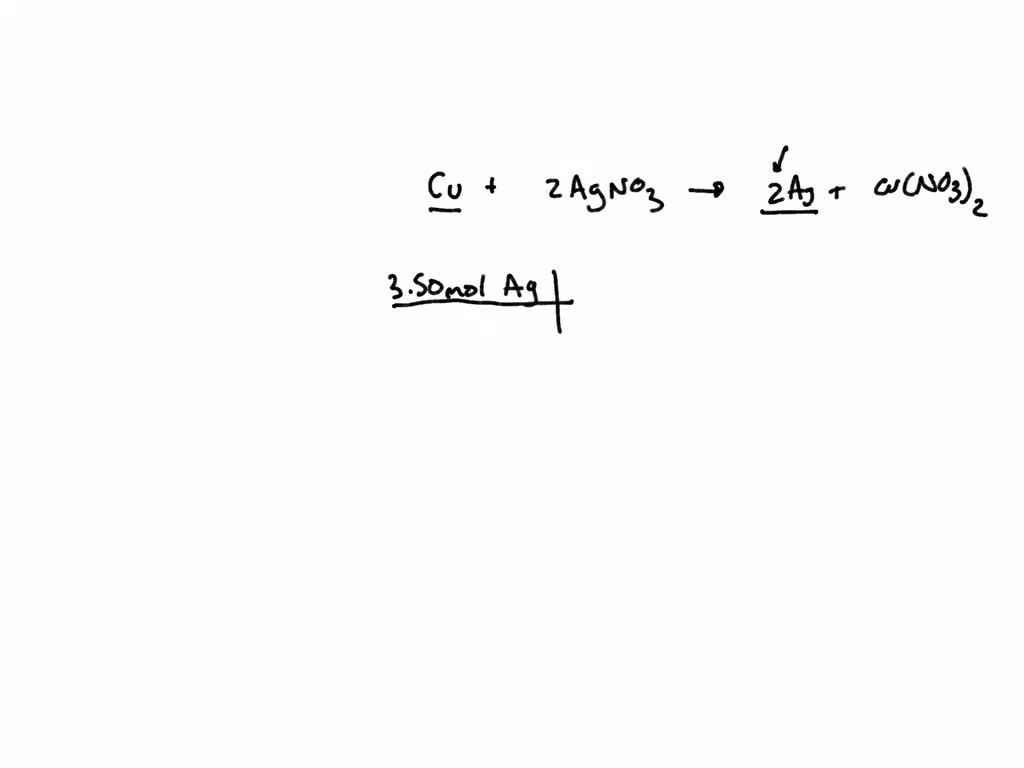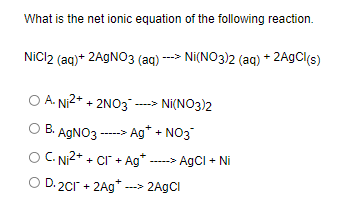A strip of nickel metal is placed in a 1 molar solution of Ni(NO3)2 and a strip of silver metal - Chemistry - Electrochemistry - 9165083 | Meritnation.com
A strip of nickel metal is placed in a 1 molar solution of Ni(NO3)2 and a strip of silver metal is placed in a one molar - Sarthaks eConnect | Largest Online